Understandings:
● Autotrophs convert carbon dioxide into carbohydrates and other carbon compounds.
● In aquatic ecosystems carbon is present as dissolved carbon dioxide and hydrogen carbonate ions.
● Carbon dioxide diffuses from the atmosphere or water into autotrophs.
● Carbon dioxide is produced by respiration and diffuses out of organisms into water or the atmosphere.
● Methane is produced from organic matter in anaerobic conditions by methanogenic archaeans and some diffuses into the atmosphere or accumulates in the ground.
● Methane is oxidized to carbon dioxide and water in the atmosphere.
● Peat forms when organic matter is not fully decomposed because of acidic and/or anaerobic conditions in waterlogged soils.
● Partially decomposed organic matter from past geological eras was converted either into coal or into oil and gas which accumulates in porous rocks.
● Carbon dioxide is produced by the combustion of biomass and fossilized organic matter.
● Animals such as reef-building corals and molluscs have hard parts that are composed of calcium carbonate and can become fossilized in limestone.
Applications and skills:
● Application: Estimation of carbon fluxes due to processes in the carbon cycle.
● Application: Analysis of data from air monitoring stations to explain annual fluctuations. ● Skill: Construct a diagram of the carbon cycle.
Guidance
● Carbon fluxes should be measured in gigatonnes.
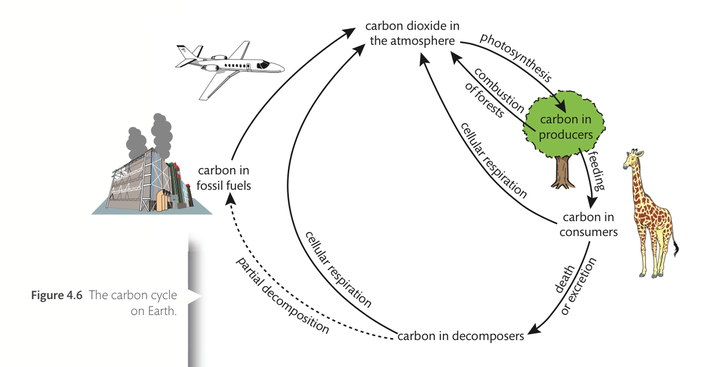
Carbon
Carbon is such a crucial element to living organisms that it is part of the definition of a living thing. You will recall that the term ‘organic’ implies that carbon is present. Hence, life on Earth is referred to as carbon-based life.
Not only is carbon found in the biosphere in organic molecules such as carbohydrates, proteins, lipids, and vitamins, it is also found in the atmosphere as carbon dioxide and in the lithosphere as carbonates and fossil fuels in rocks. The biosphere refers to all the places where life is found, and the lithosphere refers to all the places where rocks are found.
Carbon is constantly being cycled between living organisms and inorganic processes that allow the carbon to be available. The carbon atoms that make up the cells of the flesh and blood of the giraffe, for example, came from the vegetation the giraffe ate. Eating organic material provides newly dividing cells in the giraffe’s body with a fresh supply of carbon-based energy-rich molecules with which the cells can carry out work. When cellular respiration is complete, carbon dioxide is released into the atmosphere, and when the giraffe dies, its body will be eaten by scavengers and the remains broken down by decomposers. Some of the carbon from the giraffe’s body will go back into the atmosphere as carbon dioxide when the decomposers perform cellular respiration.
● Autotrophs convert carbon dioxide into carbohydrates and other carbon compounds.
● In aquatic ecosystems carbon is present as dissolved carbon dioxide and hydrogen carbonate ions.
● Carbon dioxide diffuses from the atmosphere or water into autotrophs.
● Carbon dioxide is produced by respiration and diffuses out of organisms into water or the atmosphere.
● Methane is produced from organic matter in anaerobic conditions by methanogenic archaeans and some diffuses into the atmosphere or accumulates in the ground.
● Methane is oxidized to carbon dioxide and water in the atmosphere.
● Peat forms when organic matter is not fully decomposed because of acidic and/or anaerobic conditions in waterlogged soils.
● Partially decomposed organic matter from past geological eras was converted either into coal or into oil and gas which accumulates in porous rocks.
● Carbon dioxide is produced by the combustion of biomass and fossilized organic matter.
● Animals such as reef-building corals and molluscs have hard parts that are composed of calcium carbonate and can become fossilized in limestone.
Applications and skills:
● Application: Estimation of carbon fluxes due to processes in the carbon cycle.
● Application: Analysis of data from air monitoring stations to explain annual fluctuations. ● Skill: Construct a diagram of the carbon cycle.
Guidance
● Carbon fluxes should be measured in gigatonnes.
Carbon
Carbon is such a crucial element to living organisms that it is part of the definition of a living thing. You will recall that the term ‘organic’ implies that carbon is present. Hence, life on Earth is referred to as carbon-based life.
Not only is carbon found in the biosphere in organic molecules such as carbohydrates, proteins, lipids, and vitamins, it is also found in the atmosphere as carbon dioxide and in the lithosphere as carbonates and fossil fuels in rocks. The biosphere refers to all the places where life is found, and the lithosphere refers to all the places where rocks are found.
Carbon is constantly being cycled between living organisms and inorganic processes that allow the carbon to be available. The carbon atoms that make up the cells of the flesh and blood of the giraffe, for example, came from the vegetation the giraffe ate. Eating organic material provides newly dividing cells in the giraffe’s body with a fresh supply of carbon-based energy-rich molecules with which the cells can carry out work. When cellular respiration is complete, carbon dioxide is released into the atmosphere, and when the giraffe dies, its body will be eaten by scavengers and the remains broken down by decomposers. Some of the carbon from the giraffe’s body will go back into the atmosphere as carbon dioxide when the decomposers perform cellular respiration.
The role of autotrophs in the carbon cycle
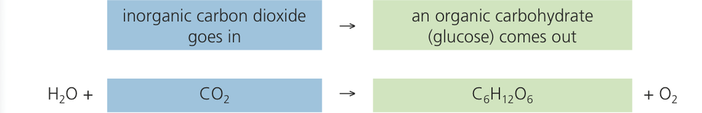
Photosynthetic autotrophs take carbon dioxide from the atmosphere and convert it into carbohydrates. Here is the unbalanced chemical equation for photosynthesis.
Photosynthetic autotrophs take carbon dioxide from the atmosphere and convert it into carbohydrates. Here is the unbalanced chemical equation for photosynthesis.
The sugar on the right-hand side of the equation (in green) is a source of food, not only to the autotroph synthesizing it, but also to the organisms that feed on the autotrophs. In its inorganic form on the left, as atmospheric carbon dioxide (in blue), the carbon is not usable as a food source by the autotrophs or by any consumers.
From the C6H12O6 molecules, autotrophs can manufacture other compounds. Fructose and galactose are other sugars that can be made by plants from glucose. Connecting the sugars together into a long chain can make starch; plants can store energy for a future season or a future generation in the form of starch granules, tubers, or seeds. Plants and algae need to build their cell walls with cellulose, which is also made from long chains of glucose. Glucose is the starting point for making other organic compounds that are not carbohydrates, such as lipids and amino acids. These compounds are necessary to make useful things such as cell membranes and proteins such as enzymes. To synthesize these non-carbohydrates, other elements such as nitrogen must be added to the glucose.
Carbon in aquatic ecosystems
As you know from drinking fizzy drinks or carbonated water, carbon dioxide can dissolve in water. Although the oceans, lakes, and rivers of the world are not as fizzy as a carbonated drink, they contain dissolved carbon dioxide because carbon dioxide from the atmosphere can be absorbed by the water. Remember also that organisms living in water produce carbon dioxide through cellular respiration. As the carbon dioxide is dissolved in the water, it forms an acid. The pH of water
decreases as the amount of carbon dioxide increases. This is why carbonated water has an acidic taste.
From the C6H12O6 molecules, autotrophs can manufacture other compounds. Fructose and galactose are other sugars that can be made by plants from glucose. Connecting the sugars together into a long chain can make starch; plants can store energy for a future season or a future generation in the form of starch granules, tubers, or seeds. Plants and algae need to build their cell walls with cellulose, which is also made from long chains of glucose. Glucose is the starting point for making other organic compounds that are not carbohydrates, such as lipids and amino acids. These compounds are necessary to make useful things such as cell membranes and proteins such as enzymes. To synthesize these non-carbohydrates, other elements such as nitrogen must be added to the glucose.
Carbon in aquatic ecosystems
As you know from drinking fizzy drinks or carbonated water, carbon dioxide can dissolve in water. Although the oceans, lakes, and rivers of the world are not as fizzy as a carbonated drink, they contain dissolved carbon dioxide because carbon dioxide from the atmosphere can be absorbed by the water. Remember also that organisms living in water produce carbon dioxide through cellular respiration. As the carbon dioxide is dissolved in the water, it forms an acid. The pH of water
decreases as the amount of carbon dioxide increases. This is why carbonated water has an acidic taste.
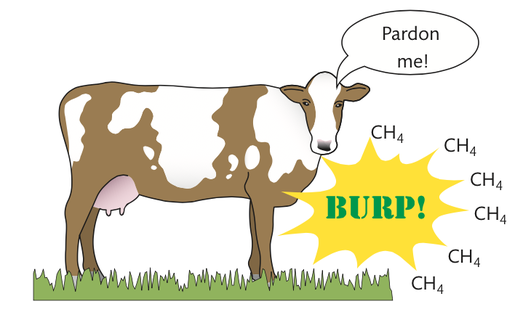
The oxidation of methane
methane burning in oxygen gas:
CH4 + 2O2 → 2H2O + CO2
Methane is the main ingredient in the fossil fuel we call natural gas. As you can see from the formula, this chemical reaction involves oxygen gas from Earth’s atmosphere. When the methane is oxidized, the two molecules produced are water vapour and carbon dioxide gas.
The carbon found in the molecule CH4 was borrowed from a CO2 molecule that was removed from the atmosphere millions of years ago during photosynthesis. It then took the methane gas millions of years to form and accumulate underground. When we burn natural gas provided by the petroleum industry, we return that carbon to the atmosphere in the form of carbon dioxide.
Peat as a fossil fuel
Another organic substance that can be used as a fossil fuel is partially decomposed plant material called peat. Peat is a kind of waterlogged soil found in certain types of wetlands, such as mires and bogs. Peat is very dark in colour and only certain types of vegetation can grow on its surface, such as sphagnum moss. Although peat is a heterogeneous mixture of many things, at least 30% of its dry mass must be composed of dead organic material for it to be called peat. The soil that forms peat is called a histosol.
The high levels of water on peatland force out the air that would normally be between the soil particles. As a result, anaerobic conditions are created, which allows certain types of microorganisms to grow but prevents the growth of microorganisms that would normally help in the decomposition of plant material. Hence many of the energy- rich molecules that would have been fed upon by decomposers are left behind and transformed, over thousands of years, into in energy-rich peat.
Another characteristic of peatlands is the pH of the waterlogged histosol: it is very acidic. Just as with low oxygen levels, if the acidity is not conducive to the decomposers, they will not be able to do their work. High acidity contributes to the fact that non-decomposed material accumulates. In the pools of acidic water that can be found on these wetlands, certain types of organisms can be found that are not found anywhere else, such as some species of aquatic beetles.
Oil and gas as fossil fuels
In some cases, when left in the correct conditions, partially decomposed peat can
be further transformed into coal. Over millions of years, sediments can accumulate above the peat, and the weight and pressure of those sediments compresses the peat. Under conditions ideal for the formation of coal, the sedimentation continues until the carbon-rich deposits are not only under huge pressure but also exposed to high temperatures because they have been pushed far below Earth’s surface. The pressure and heat cause chemical transformations associated with lithification, which is the transformation of sediments into solid hydrogen atoms rock. During lithification, the molecules H are compacted and rearranged.
What is of great interest to industries using coal is the hydrocarbons, the long chains of carbon atoms attached to hydrogen atoms.
The C–H bonds hold a significant amount of energy, and, because there are many of them in long chains, each hydrocarbon molecule is rich in energy ready to be released by burning.
In order to use coal for energy, it must be extracted from below the ground, which is why mining is necessary. Coal is found in seams, where the layers of sediments were deposited, covered, and then transformed and often twisted and deformed by geological forces over millions of years.
In addition to coal, chemical transformations underground can produce other petroleum products, such as crude oil and, as we have seen, natural gas.
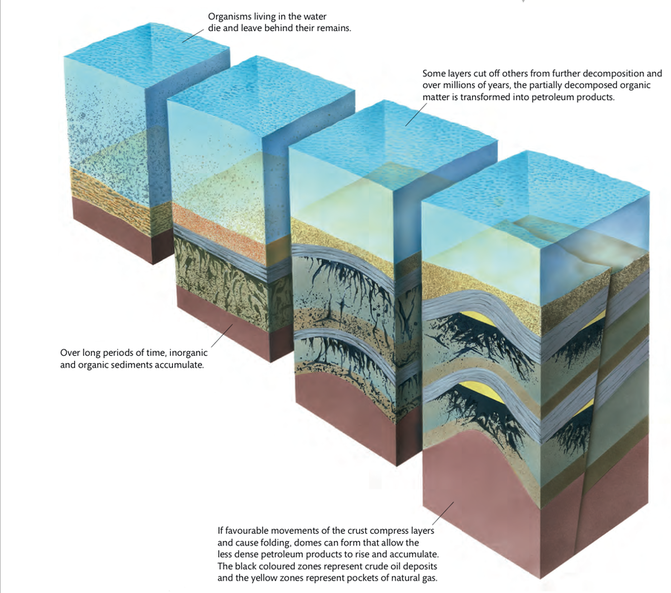
For this, we have to go far back in time, before dinosaurs roamed Earth. During the Carboniferous period, hundreds of millions of years ago, some places in the world that are now dry land were underwater and hosted abundant aquatic or marine life, including algae and zooplankton. For example, the dry deserts of Saudi Arabia used to be under the Tethys Ocean, back when all the continents were still stuck together in the supercontinent called Pangaea.
At that distant time in Earth’s past, under conditions ideal for the formation of petroleum products, the dead remains of the organisms in the water did not fully decompose at the bottom of the ocean, and instead formed layers of sediment along with silt. In conditions lacking oxygen (anoxic conditions), the decaying material started to form sludge, as some parts of the organisms’ cells decayed while others did not. One component of dead algae and zooplankton that is not easily broken down is the lipid component of their cells. Accumulated lipids that are trapped in sediments at the bottom of an ocean form a waxy substance called kerogen. It, too, is rich in hydrocarbons and, like the formation of other fossil fuels, is transformed by pressure and heat as sediments accumulate above it and cause its molecules to rearrange.
Over millions of years, and after geological transformation, the kerogen in porous sedimentary rock becomes crude oil or, if it is in a gas state, natural gas. Both of these petroleum products are less dense than rock, so they tend to rise through cracks in the rocks towards the surface.
methane burning in oxygen gas:
CH4 + 2O2 → 2H2O + CO2
Methane is the main ingredient in the fossil fuel we call natural gas. As you can see from the formula, this chemical reaction involves oxygen gas from Earth’s atmosphere. When the methane is oxidized, the two molecules produced are water vapour and carbon dioxide gas.
The carbon found in the molecule CH4 was borrowed from a CO2 molecule that was removed from the atmosphere millions of years ago during photosynthesis. It then took the methane gas millions of years to form and accumulate underground. When we burn natural gas provided by the petroleum industry, we return that carbon to the atmosphere in the form of carbon dioxide.
Peat as a fossil fuel
Another organic substance that can be used as a fossil fuel is partially decomposed plant material called peat. Peat is a kind of waterlogged soil found in certain types of wetlands, such as mires and bogs. Peat is very dark in colour and only certain types of vegetation can grow on its surface, such as sphagnum moss. Although peat is a heterogeneous mixture of many things, at least 30% of its dry mass must be composed of dead organic material for it to be called peat. The soil that forms peat is called a histosol.
The high levels of water on peatland force out the air that would normally be between the soil particles. As a result, anaerobic conditions are created, which allows certain types of microorganisms to grow but prevents the growth of microorganisms that would normally help in the decomposition of plant material. Hence many of the energy- rich molecules that would have been fed upon by decomposers are left behind and transformed, over thousands of years, into in energy-rich peat.
Another characteristic of peatlands is the pH of the waterlogged histosol: it is very acidic. Just as with low oxygen levels, if the acidity is not conducive to the decomposers, they will not be able to do their work. High acidity contributes to the fact that non-decomposed material accumulates. In the pools of acidic water that can be found on these wetlands, certain types of organisms can be found that are not found anywhere else, such as some species of aquatic beetles.
Oil and gas as fossil fuels
In some cases, when left in the correct conditions, partially decomposed peat can
be further transformed into coal. Over millions of years, sediments can accumulate above the peat, and the weight and pressure of those sediments compresses the peat. Under conditions ideal for the formation of coal, the sedimentation continues until the carbon-rich deposits are not only under huge pressure but also exposed to high temperatures because they have been pushed far below Earth’s surface. The pressure and heat cause chemical transformations associated with lithification, which is the transformation of sediments into solid hydrogen atoms rock. During lithification, the molecules H are compacted and rearranged.
What is of great interest to industries using coal is the hydrocarbons, the long chains of carbon atoms attached to hydrogen atoms.
The C–H bonds hold a significant amount of energy, and, because there are many of them in long chains, each hydrocarbon molecule is rich in energy ready to be released by burning.
In order to use coal for energy, it must be extracted from below the ground, which is why mining is necessary. Coal is found in seams, where the layers of sediments were deposited, covered, and then transformed and often twisted and deformed by geological forces over millions of years.
In addition to coal, chemical transformations underground can produce other petroleum products, such as crude oil and, as we have seen, natural gas.
For this, we have to go far back in time, before dinosaurs roamed Earth. During the Carboniferous period, hundreds of millions of years ago, some places in the world that are now dry land were underwater and hosted abundant aquatic or marine life, including algae and zooplankton. For example, the dry deserts of Saudi Arabia used to be under the Tethys Ocean, back when all the continents were still stuck together in the supercontinent called Pangaea.
At that distant time in Earth’s past, under conditions ideal for the formation of petroleum products, the dead remains of the organisms in the water did not fully decompose at the bottom of the ocean, and instead formed layers of sediment along with silt. In conditions lacking oxygen (anoxic conditions), the decaying material started to form sludge, as some parts of the organisms’ cells decayed while others did not. One component of dead algae and zooplankton that is not easily broken down is the lipid component of their cells. Accumulated lipids that are trapped in sediments at the bottom of an ocean form a waxy substance called kerogen. It, too, is rich in hydrocarbons and, like the formation of other fossil fuels, is transformed by pressure and heat as sediments accumulate above it and cause its molecules to rearrange.
Over millions of years, and after geological transformation, the kerogen in porous sedimentary rock becomes crude oil or, if it is in a gas state, natural gas. Both of these petroleum products are less dense than rock, so they tend to rise through cracks in the rocks towards the surface.
In order to be used by humans, petroleum products must be trapped and pooled under a non-porous rock, preferably one that is bent by tectonic movement into a dome, as seen in Figure 4.12. This kind of formation allows large quantities of useful gas and oil to collect together in a productive reservoir. Geologists study the porosity and deformations of rock layers in order to determine which parts of the world might contain exploitable gas and oil reserves.
Carbon dioxide is produced when fossil fuels are used
Substances rich in hydrocarbons can be oxidized using oxygen gas from the atmosphere when they are burned. If you have ever made a fire on a beach or at a campsite, you know that organic material such as wood is capable of releasing a considerable amount of energy in the form of light and heat. Wood is not the only fuel of biological origin that can be burned: many people living in non-industrialized areas of the world use biomass in the form of animal dung as a source of energy. The dried dung of domesticated animals such as cows can be burned and used for various purposes, including cooking. Fresh, wet dung can be mixed with other refuse from a farm and put into a large container, where methane- producing microorganisms will decompose and ferment the material to produce flammable methane gas
In an effort to reduce fossil fuel consumption, some countries, such as the USA and Brazil, have introduced biofuel programmes using ethanol made from crops such as corn and soybeans. The plant material is fed to microorganisms that ferment it and in the process release ethanol. The ethanol is added to gasoline for vehicles, and contributes to a reduction in gasoline use. Standard vehicles cannot use more than 25% ethanol and need 75% or more gasoline (this mix can also be called gasohol), but vehicles specially adapted for biofuels can run solely on ethanol.
Using a different technique, biodiesel can be made from vegetable oils or animal fat. Some people have even modified their cars so that they run on the waste oil from deep-fat fryers at fast food restaurants.
Although it can be argued that using biofuels allows countries to reduce their dependence on imported fossil fuels, the burning of any biomass still releases carbon dioxide into the atmosphere. The difference is that, unlike fossil fuels, the carbon dioxide from biofuels was removed from the atmosphere by plants just a few months or years before the biofuel was used.
Limestone
Marine organisms take dissolved carbon out of the water and use some of it to make their carbonate shells. Carbon can be in the form of carbon dioxide dissolved in the water or it can be in the form of hydrogen carbonate ions. The organisms that build coral reefs are called coral polyps, and they absorb two ions from the seawater to build the reef: hydrogen carbonate ions and calcium ions. When combined, molecules of calcium carbonate (CaCO3) are formed. This molecule is the basis of the coral reef, and it is sturdy like rock. Below is the chemical equation for making calcium carbonate:
Ca2+ + 2 HCO3– → CaCO3 + CO2 + H2O
Other organisms use calcium carbonate to build shells around their bodies. Molluscs build up their shells with calcium carbonate and, when they die, the shells accumulate at the bottom of the ocean.
Microscopic foraminifera usually live on the ocean floor and are also very good at building shells, albeit very small ones. Because they are so numerous, however, and they have been around for hundreds of millions of years, their shells have accumulated in sediments, and when the sediments go through the process of lithification, they form limestone. Limestone has long been used by humans as a building material and is a major ingredient in modern cement.
The process of taking carbon out of the environment and ‘locking it up’ in a substance for an extended period of time is called carbon sequestration, and when it happens naturally it is called biosequestration. This is one way balance is maintained in the carbon cycle.
Carbon dioxide is produced when fossil fuels are used
Substances rich in hydrocarbons can be oxidized using oxygen gas from the atmosphere when they are burned. If you have ever made a fire on a beach or at a campsite, you know that organic material such as wood is capable of releasing a considerable amount of energy in the form of light and heat. Wood is not the only fuel of biological origin that can be burned: many people living in non-industrialized areas of the world use biomass in the form of animal dung as a source of energy. The dried dung of domesticated animals such as cows can be burned and used for various purposes, including cooking. Fresh, wet dung can be mixed with other refuse from a farm and put into a large container, where methane- producing microorganisms will decompose and ferment the material to produce flammable methane gas
In an effort to reduce fossil fuel consumption, some countries, such as the USA and Brazil, have introduced biofuel programmes using ethanol made from crops such as corn and soybeans. The plant material is fed to microorganisms that ferment it and in the process release ethanol. The ethanol is added to gasoline for vehicles, and contributes to a reduction in gasoline use. Standard vehicles cannot use more than 25% ethanol and need 75% or more gasoline (this mix can also be called gasohol), but vehicles specially adapted for biofuels can run solely on ethanol.
Using a different technique, biodiesel can be made from vegetable oils or animal fat. Some people have even modified their cars so that they run on the waste oil from deep-fat fryers at fast food restaurants.
Although it can be argued that using biofuels allows countries to reduce their dependence on imported fossil fuels, the burning of any biomass still releases carbon dioxide into the atmosphere. The difference is that, unlike fossil fuels, the carbon dioxide from biofuels was removed from the atmosphere by plants just a few months or years before the biofuel was used.
Limestone
Marine organisms take dissolved carbon out of the water and use some of it to make their carbonate shells. Carbon can be in the form of carbon dioxide dissolved in the water or it can be in the form of hydrogen carbonate ions. The organisms that build coral reefs are called coral polyps, and they absorb two ions from the seawater to build the reef: hydrogen carbonate ions and calcium ions. When combined, molecules of calcium carbonate (CaCO3) are formed. This molecule is the basis of the coral reef, and it is sturdy like rock. Below is the chemical equation for making calcium carbonate:
Ca2+ + 2 HCO3– → CaCO3 + CO2 + H2O
Other organisms use calcium carbonate to build shells around their bodies. Molluscs build up their shells with calcium carbonate and, when they die, the shells accumulate at the bottom of the ocean.
Microscopic foraminifera usually live on the ocean floor and are also very good at building shells, albeit very small ones. Because they are so numerous, however, and they have been around for hundreds of millions of years, their shells have accumulated in sediments, and when the sediments go through the process of lithification, they form limestone. Limestone has long been used by humans as a building material and is a major ingredient in modern cement.
The process of taking carbon out of the environment and ‘locking it up’ in a substance for an extended period of time is called carbon sequestration, and when it happens naturally it is called biosequestration. This is one way balance is maintained in the carbon cycle.