Understandings:
● Nitrogen-fixing bacteria convert atmospheric nitrogen to ammonia.
● Rhizobium associates with roots in a mutualistic relationship.
● In the absence of oxygen, denitrifying bacteria reduce nitrate in the soil.
● Phosphorous can be added to the phosphorous cycle by application of fertilizer, or removed by the harvesting of agricultural crops.
● The rate of turnover in the phosphorous cycle is much lower than the nitrogen cycle.
● Availability of phosphorous may become limiting to agriculture in the future.
● Leaching of mineral nutrients from agricultural land into rivers causes eutrophication and leads to increased biochemical oxygen demand.
Applications and skills:
● Application: The impact of waterlogging on the nitrogen cycle.
● Application: Insectivorous plants as an adaptation for low nitrogen availability in waterlogged soils.
● Skills: Drawing and labelling a diagram of the nitrogen cycle.
● Skills: Assess the nutrient content of a soil sample.
The nitrogen cycle
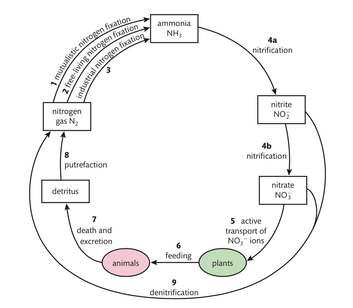
Bacteria play a hugely important part in the processes by which nitrogen is continuously recycled through the environment. Roles of bacteria in the nitrogen cycle are summarized in the following figure:
● Nitrogen-fixing bacteria convert atmospheric nitrogen to ammonia.
● Rhizobium associates with roots in a mutualistic relationship.
● In the absence of oxygen, denitrifying bacteria reduce nitrate in the soil.
● Phosphorous can be added to the phosphorous cycle by application of fertilizer, or removed by the harvesting of agricultural crops.
● The rate of turnover in the phosphorous cycle is much lower than the nitrogen cycle.
● Availability of phosphorous may become limiting to agriculture in the future.
● Leaching of mineral nutrients from agricultural land into rivers causes eutrophication and leads to increased biochemical oxygen demand.
Applications and skills:
● Application: The impact of waterlogging on the nitrogen cycle.
● Application: Insectivorous plants as an adaptation for low nitrogen availability in waterlogged soils.
● Skills: Drawing and labelling a diagram of the nitrogen cycle.
● Skills: Assess the nutrient content of a soil sample.
The nitrogen cycle
Bacteria play a hugely important part in the processes by which nitrogen is continuously recycled through the environment. Roles of bacteria in the nitrogen cycle are summarized in the following figure:
- Mutualistic nitrogen fixation. Certain bacteria form a symbiotic relationship with a host plant and fix nitrogen for it, e.g. Rhizobium lives in symbiosis with legumes (beans, peas, and clover).
- Free-living nitrogen fixation. Nitrogen-fixing bacteria, e.g. Azotobacter, live freely in the soil and do not need a host.
- Industrial nitrogen fixation. Burning fossil fuels to produce fertilizer is an important source of fixed nitrogen.
- a) Nitrification. Oxygen is needed to turn ammonia into nitrites by bacteria in the soil, e.g. Nitrosomonas. b) Nitrification. Oxygen is also required to change nitrites into nitrates by soil bacteria, e.g. Nitrobacter.
- Active transport of nitrates. Nitrates are actively transported by plants (using ATP for energy) into their roots.
- Plants and animals. Plants use nitrates to make their own proteins. This process is called assimilation. Animals feed on plants, digest, and rearrange plant proteins to make their own proteins.
- Death and excretion. The waste products of digestion and dead bodies of plants and animals are full of molecules containing nitrogen.
- Putrefaction. Decomposers such as bacteria and fungi break down complex proteins, and release nitrogen gas into the atmosphere.
- Denitrification. Some bacteria, e.g. Pseudomonas denitrificans, remove nitrates and nitrites, and put nitrogen gas back into the atmosphere.
Conditions that favour nitrification and denitrification
Nitrification occurs as a result of the actions of two bacteria. These are aerobic reactions carried out by two autotrophic bacteria that are beneficial to the environment. The conditions required for nitrification are:
• available oxygen (the reaction is aerobic)
• neutral pH (preferred by the two bacteria)
• warm temperature (preferred by the two bacteria).
Denitrification is the conversion of nitrates to nitrogen gas. This takes place in anaerobic conditions by autotrophic bacteria. The conditions required for denitrification are:
• no available oxygen (e.g. flooding or compacted soil)
• a high nitrogen input.
A product of denitrification, nitrous oxide (NO), can contribute to global warming, as it is a minor greenhouse gas.
Release of nitrate fertilizer into rivers
The presence of excess nitrates and phosphates in rivers and streams is termed eutrophication. The process of eutrophication proceeds as follows:
- high nitrates and phosphates fertilize the algae present in water
- there is increased growth of algae (called an algal bloom)
- the algae are decomposed by aerobic bacteria, which use up the oxygen in the water (a high use of oxygen is called biochemical oxygen demand, BOD)
- the water becomes low in oxygen (deoxygenation), and fish and other organisms that need oxygen die.
Insectivorous plants in waterlogged soils with low nitrogen
Plants need nitrogen for protein synthesis and to make new DNA molecules. Insectivorous plants such as the pitcher plant derive their nitrogen from trapping and consuming insects. Pitcher plants are adapted to living in waterlogged areas where nitrogen is lacking. Pitcher plants also need a large amount of the nitrogen-rich enzyme rubisco. This enzyme is involved in the first major step of making glucose during photosynthesis
Impact of waterlogging on the nitrogen cycle
Healthy plant roots need oxygen. Soil that is waterlogged is so saturated with water that oxygen cannot get into the soil. Waterlogged soils create anaerobic soil conditions with no oxygen. Anaerobic conditions facilitate the growth of denitrifying bacteria, which convert the nitrates needed by plants back to gaseous nitrogen. Waterlogged soils become nitrate depleted and plant (crop) growth is reduced significantly. Waterlogging can interfere with the normal cycle of nitrogen moving from the atmosphere to plants as nitrates and then back to the atmosphere.
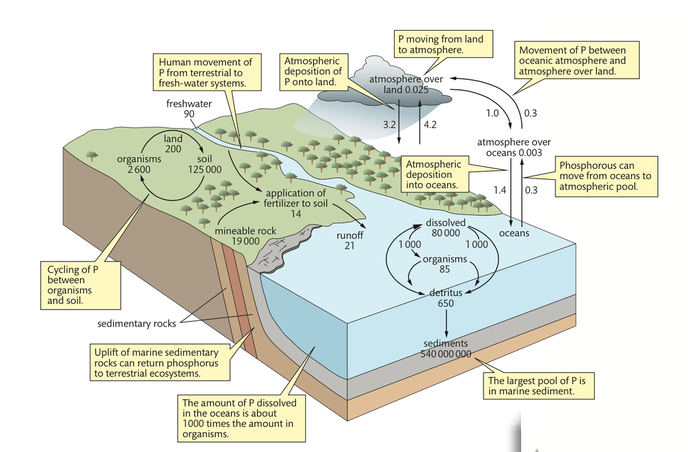
The phosphorous cycle
Phosphorous is an essential element in living systems. For example, phosphorous forms part of ATP, RNA, DNA, and phospholipid molecules. Phosphorous is not very abundant in the biosphere and there is not a substantial atmospheric pool of phosphorous as there is for carbon and nitrogen. The largest quantity of phosphorous is found in marine sediments and mineral deposits. Sedimentary rocks that are rich in phosphorous are mined for fertilizer and applied to soils. When crops are harvested, the phosphorous is removed. The only method of replacing it again is to add more fertilizer. Unlike nitrogen, which can be fixed by bacteria and added back to the soil, phosphorous is not recycled easily. Composting is one method of recycling phosphorus; however, composting is not easily done on a large scale.
The rate of turnover in the phosphorous cycle is low
Phosphorous is slowly released into ecosystems from weathering rocks. As it is released it can be absorbed by the roots of plants or washed into rivers. The phosphorous that is washed away eventually finds its way into oceans, where it remains in a dissolved form until finding its way into ocean sediments. The sediments eventually form sedimentary rocks. Sedimentary rocks that slowly wear away make phosphorus available again to plants.
The availability of phosphorous may become limiting to agriculture
Currently, the major use of phosphate is in chemical fertilizer, which is used in modern agricultural production. Mining phosphorous for fertilizer is consuming the phosphorus more quickly than geological cycles can replace it, and crops remove phosphate from the soil. Most of the world’s farms do not have enough phosphate. Phosphate reserves are limited, and it is estimated that the world phosphate reserves will only last another 50–100 years. Feeding the world’s increasing population requires more and more crop production, and more and more phosphate. This is accelerating the rate of the depletion of phosphate reserves.
Currently, the major use of phosphate is in chemical fertilizer, which is used in modern agricultural production. Mining phosphorous for fertilizer is consuming the phosphorus more quickly than geological cycles can replace it, and crops remove phosphate from the soil. Most of the world’s farms do not have enough phosphate. Phosphate reserves are limited, and it is estimated that the world phosphate reserves will only last another 50–100 years. Feeding the world’s increasing population requires more and more crop production, and more and more phosphate. This is accelerating the rate of the depletion of phosphate reserves.