Applications: Urea as en example of a
compound that is produced by living organisms but can also be artificially
synthesized
Skills: - Drawing molecular diagrams of glucose, ribose, a saturated fatty acid, and a generalized amino acid. – Identification of biochemicals such as carbohydrate, lipid, or protein from molecular diagrams
Understanding:
- Molecular biology explains living processes in terms of the chemical substances involved.
- Carbon atoms can form four bonds allowing a diversity of compounds to exist
- Life if based on carbon compounds including carbohydrates, lipids, proteins and nucleic acids
- Metabolism is the web of all enzyme-catalyzed reactions in a cell or organism
- Anabolism is the synthesis of complex molecules from simple molecules including the formation of macromolecules from monomers by condensation reactions
- Catabolism is the breakdown of complex molecules into simpler molecules including the hydrolysis of macromolecules into monomers.
Molecular biology explains living processes in the terms of the chemical substances involved.
- Carbon Compounds: it is the 15th most abundant element on earth, but it can be used to make a huge range of different molecules. Carbon atoms form covalent bonds with other atoms. A covalent bond is formed when two adjacent atoms share electrons, with one electron contributed by each atom. Each carbon atom can form up to four covalent bonds, more than most other atoms, so molecules with carbon atoms can have complex structures. Carbon atoms can bond with other carbon atoms to make rings or chains of any length. Fatty acids are an example of this because they contain chains of up to 20 carbon atoms. Bonds can also be found with other elements such as hydrogen, oxygen, nitrogen and phosphorus.
- Classifying Carbon compounds: Life is based on the four main carbon compounds which are: carbohydrates, proteins, lipids and nucleic acids. Each has different properties and thus different purposes.
- Carbohydrates are composed of carbon, hydrogen and oxygen, with hydrogen and oxygen in the ratio of two hydrogen atoms to one oxygen. That is where the name carbohydrate comes from.
- Proteins: they are composed of one or more chains of amino acids, and each of these contain carbon, hydrogen, oxygen, and nitrogen.
- Nucleic acids: these are the chains of subunits called nucleotides, which contain carbon, hydrogen, oxygen, nitrogen, and phosphorus. There are two types of nucleic acids: RNA (ribonucleic acid) and DNA (deoxyribonucleic acid)
Drawing molecular diagrams for glucose, ribose, a saturated fatty acid and a generalized amino acid.
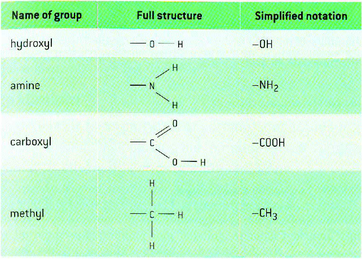
Chemical groups:
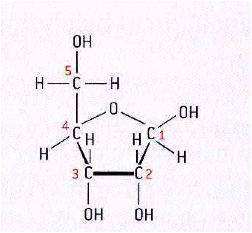
- Ribose:
* The formula of ribose is C5H10O5
* The molecules is a five-membered ring with a side chain
* four carbon atoms are in the ring and one forms the side chain
* the carbon atoms can be numbered starting with number 1 on the right
*the hydroxyl groups (OH) on carbon atoms 1, 2 and 3 point up, down and down.
Skills: - Drawing molecular diagrams of glucose, ribose, a saturated fatty acid, and a generalized amino acid. – Identification of biochemicals such as carbohydrate, lipid, or protein from molecular diagrams
Understanding:
- Molecular biology explains living processes in terms of the chemical substances involved.
- Carbon atoms can form four bonds allowing a diversity of compounds to exist
- Life if based on carbon compounds including carbohydrates, lipids, proteins and nucleic acids
- Metabolism is the web of all enzyme-catalyzed reactions in a cell or organism
- Anabolism is the synthesis of complex molecules from simple molecules including the formation of macromolecules from monomers by condensation reactions
- Catabolism is the breakdown of complex molecules into simpler molecules including the hydrolysis of macromolecules into monomers.
Molecular biology explains living processes in the terms of the chemical substances involved.
- Carbon Compounds: it is the 15th most abundant element on earth, but it can be used to make a huge range of different molecules. Carbon atoms form covalent bonds with other atoms. A covalent bond is formed when two adjacent atoms share electrons, with one electron contributed by each atom. Each carbon atom can form up to four covalent bonds, more than most other atoms, so molecules with carbon atoms can have complex structures. Carbon atoms can bond with other carbon atoms to make rings or chains of any length. Fatty acids are an example of this because they contain chains of up to 20 carbon atoms. Bonds can also be found with other elements such as hydrogen, oxygen, nitrogen and phosphorus.
- Classifying Carbon compounds: Life is based on the four main carbon compounds which are: carbohydrates, proteins, lipids and nucleic acids. Each has different properties and thus different purposes.
- Carbohydrates are composed of carbon, hydrogen and oxygen, with hydrogen and oxygen in the ratio of two hydrogen atoms to one oxygen. That is where the name carbohydrate comes from.
- Proteins: they are composed of one or more chains of amino acids, and each of these contain carbon, hydrogen, oxygen, and nitrogen.
- Nucleic acids: these are the chains of subunits called nucleotides, which contain carbon, hydrogen, oxygen, nitrogen, and phosphorus. There are two types of nucleic acids: RNA (ribonucleic acid) and DNA (deoxyribonucleic acid)
Drawing molecular diagrams for glucose, ribose, a saturated fatty acid and a generalized amino acid.
Chemical groups:
- Ribose:
* The formula of ribose is C5H10O5
* The molecules is a five-membered ring with a side chain
* four carbon atoms are in the ring and one forms the side chain
* the carbon atoms can be numbered starting with number 1 on the right
*the hydroxyl groups (OH) on carbon atoms 1, 2 and 3 point up, down and down.
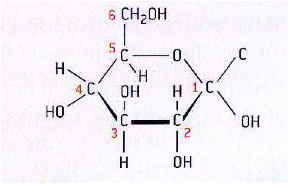
- Glucose
*the formula of glucose is C6H12O6
*five carbon atoms are in the ring and one forms the side chain
*the carbon atoms can be numbered starting with number 1 on the right
* The hydroxyl groups (OH) on carbon atoms 1, 2, 3 and 4 point down, down, up and down respectively, although in a form of glucose used by plants to make cellulose the hydroxyl group on carbon atom 1 points upwards
*the formula of glucose is C6H12O6
*five carbon atoms are in the ring and one forms the side chain
*the carbon atoms can be numbered starting with number 1 on the right
* The hydroxyl groups (OH) on carbon atoms 1, 2, 3 and 4 point down, down, up and down respectively, although in a form of glucose used by plants to make cellulose the hydroxyl group on carbon atom 1 points upwards
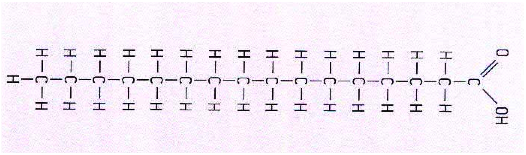
- Saturated Fatty acids
* The carbon atoms form an unbranched chain
* In saturated fatty acids they are bonded to each other by single bonds
*The number of carbon atoms is most commonly between 14 and 20
*At on end of the chain the carbon atoms is part of a carboxyl group
*At the other end the carbon atom is bonded to three hydrogen atoms
* All other carbon atoms are bonded to two hydrogen atoms
* The carbon atoms form an unbranched chain
* In saturated fatty acids they are bonded to each other by single bonds
*The number of carbon atoms is most commonly between 14 and 20
*At on end of the chain the carbon atoms is part of a carboxyl group
*At the other end the carbon atom is bonded to three hydrogen atoms
* All other carbon atoms are bonded to two hydrogen atoms
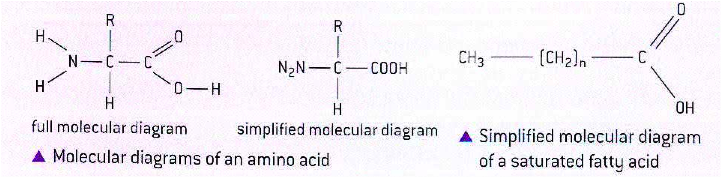
- Amino acids:
* A carbon atom in the centre of the molecule is bonded to four different things:
+ An amine group, hence the term amino acid
+A carboxyl group which makes the molecule and acid
+ A hydrogen atom
+ The R group, which is the variable part of amino acids
* A carbon atom in the centre of the molecule is bonded to four different things:
+ An amine group, hence the term amino acid
+A carboxyl group which makes the molecule and acid
+ A hydrogen atom
+ The R group, which is the variable part of amino acids
Identifying
molecules
Identification of biochemicals as carbohydrate, lipid or protein from molecular diagrams
It is usually easy to recognize each one because they are particularly different.
- Proteins contain C, H, O and N whereas carbohydrates and lipids contain C, H and O but not N
- Only proteins contain sulphur (s) in many cases
- Carbs contain hydrogen and oxygen atoms in the ratio of 2:1, for example glucose is C6H12O6 and sucrose is C12H22012
- Lipids contain less oxygen than carbs, for example, an oleic acid (unsaturated fatty acid) is C18H3502 and the steroid testosterone is C19H2802
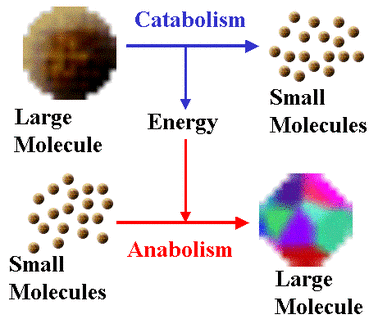
Metabolism
It is the web of all enzyme-catalyzed reactions in a cell or organism
All living organisms carry out a large number of chemical reactions, most of them occur in the cytoplasm of the cell, but there are some extracellular ones, like the breaking of food into the small intestine called digestion. Metabolism is the sum of all chemical reactions that occur within an organism.
Metabolism consists of pathways by which a type of molecule is transformed into another molecule in a series of small steps. These pathways are mostly chains of reactions but they are also in the form cycles.
Metabolism is divided into two parts:
- Anabolism
Synthesis of complex molecules from simpler molecules including the formation of macromolecules from monomers by condensation reactions
The reactions from anabolism are the building up of large molecules from smaller ones. An example could be anabolic steroids, which are hormones that promote bodybuilding. Anabolic reactions require energy in the form of ATP
Processes of anabolism:
+ Protein synthesis using ribosomes
+DNA synthesis during replication
+Photosynthesis, including production of glucose from carbon dioxide and water
+ Synthesis of complex carbs including starch, cellulose and glycogen
- Catabolism
Breakdown of complex molecules into simpler molecules including the hydrolysis (hydro=water, lysis= extraction) of macromolecules into monomers.
Part of metabolism in which larger molecules are broken down into smaller ones. Catabolic reactions release energy and in some cases it is in the form of ATP, which can then be used in the cell. Catabolism processes:
+ Digestion of food in the mouth, stomach and small intestine
+ Cell respiration in which glucose or lipids are oxidized to carbon dioxide and water
+ Digestion of complex carbon compounds in dead organic matter by decomposers
Identification of biochemicals as carbohydrate, lipid or protein from molecular diagrams
It is usually easy to recognize each one because they are particularly different.
- Proteins contain C, H, O and N whereas carbohydrates and lipids contain C, H and O but not N
- Only proteins contain sulphur (s) in many cases
- Carbs contain hydrogen and oxygen atoms in the ratio of 2:1, for example glucose is C6H12O6 and sucrose is C12H22012
- Lipids contain less oxygen than carbs, for example, an oleic acid (unsaturated fatty acid) is C18H3502 and the steroid testosterone is C19H2802
Metabolism
It is the web of all enzyme-catalyzed reactions in a cell or organism
All living organisms carry out a large number of chemical reactions, most of them occur in the cytoplasm of the cell, but there are some extracellular ones, like the breaking of food into the small intestine called digestion. Metabolism is the sum of all chemical reactions that occur within an organism.
Metabolism consists of pathways by which a type of molecule is transformed into another molecule in a series of small steps. These pathways are mostly chains of reactions but they are also in the form cycles.
Metabolism is divided into two parts:
- Anabolism
Synthesis of complex molecules from simpler molecules including the formation of macromolecules from monomers by condensation reactions
The reactions from anabolism are the building up of large molecules from smaller ones. An example could be anabolic steroids, which are hormones that promote bodybuilding. Anabolic reactions require energy in the form of ATP
Processes of anabolism:
+ Protein synthesis using ribosomes
+DNA synthesis during replication
+Photosynthesis, including production of glucose from carbon dioxide and water
+ Synthesis of complex carbs including starch, cellulose and glycogen
- Catabolism
Breakdown of complex molecules into simpler molecules including the hydrolysis (hydro=water, lysis= extraction) of macromolecules into monomers.
Part of metabolism in which larger molecules are broken down into smaller ones. Catabolic reactions release energy and in some cases it is in the form of ATP, which can then be used in the cell. Catabolism processes:
+ Digestion of food in the mouth, stomach and small intestine
+ Cell respiration in which glucose or lipids are oxidized to carbon dioxide and water
+ Digestion of complex carbon compounds in dead organic matter by decomposers