Understanding: - Monosaccharide monomers
are linked together by condensation reactions to form disaccharides and
polysaccharide polymers. – Fatty acids can be saturated, monounsaturated, or
polyunsaturated. – Unsaturated fatty acids can be cis or trans isomers. –
Triglycerides are formed by condensation from three fatty acids and one
glycerol
Applications: - Structure and function of cellulose and starch in plants and glycogen in humans. – Scientific evidence for health risks of trans-fats and saturated fats. – Lipids are more suitable for long-term energy storage in humans than carbohydrates. – Evaluation of evidence and the methods used to obtain evidence for health claims made about lipids.
Skills: -Determination of body mass index (BMI) by calculation or use of a nomogram. (Body weight in Kilos/ (height in meters)^2)
Carbohydrates
Monossacharide monomers are linked together by condensation reactions to form disaccharides and polysaccharides polymers.
Glucose, fructose and ribose are examples of monosscharides. Monossacharides can be linked together to form larger molecules.
- Monosaccharides are single sugar units
- Disaccharides consist of two monosaccharides linked together. For example maltose, which is made by the linking of two glucose molecules. Linking glucose with a fructose makes sucrose.
- Polysaccharides consist of many monosoccharides linked together. Starch, glycogen and cellulose are examples, since they are all made of the linking of various glucose molecules.
Monosaccharides combine through the process of condensation, which involves a loss of an OH (hydroxyl group) and an H from another molecule, thus forming a H20. Therefore, condensation produces water.
Linking together monosaccharides to form disaccharides and polysaccharides is an anabolic process and energy has to be used to do it. ATP supplies energy to the monosaccharides and this energy is then used for condensation.
Applications: - Structure and function of cellulose and starch in plants and glycogen in humans. – Scientific evidence for health risks of trans-fats and saturated fats. – Lipids are more suitable for long-term energy storage in humans than carbohydrates. – Evaluation of evidence and the methods used to obtain evidence for health claims made about lipids.
Skills: -Determination of body mass index (BMI) by calculation or use of a nomogram. (Body weight in Kilos/ (height in meters)^2)
Carbohydrates
Monossacharide monomers are linked together by condensation reactions to form disaccharides and polysaccharides polymers.
Glucose, fructose and ribose are examples of monosscharides. Monossacharides can be linked together to form larger molecules.
- Monosaccharides are single sugar units
- Disaccharides consist of two monosaccharides linked together. For example maltose, which is made by the linking of two glucose molecules. Linking glucose with a fructose makes sucrose.
- Polysaccharides consist of many monosoccharides linked together. Starch, glycogen and cellulose are examples, since they are all made of the linking of various glucose molecules.
Monosaccharides combine through the process of condensation, which involves a loss of an OH (hydroxyl group) and an H from another molecule, thus forming a H20. Therefore, condensation produces water.
Linking together monosaccharides to form disaccharides and polysaccharides is an anabolic process and energy has to be used to do it. ATP supplies energy to the monosaccharides and this energy is then used for condensation.
Lipids
Triglycerides are formed by condensation from three fatty acids and one glycerol.
Lipids are known for being insoluble in water. Triglycerides are one of the principal groups of lipids. Examples are the fat in adipose tissue in humans and oil in sunflower seeds. Fats are liquid at body temperature but solid at room temperature, whereas oils are liquid at both temperatures.
A triglyceride is made from combining three fatty acids and one glycerol through the process of condensation, meaning that three water molecules are produced. The linkage between each fatty acid and the glycerol is an ester bond. Triglycerides are used as energy stores. The energy from them can be released by aerobic cell respiration.
Triglycerides are formed by condensation from three fatty acids and one glycerol.
Lipids are known for being insoluble in water. Triglycerides are one of the principal groups of lipids. Examples are the fat in adipose tissue in humans and oil in sunflower seeds. Fats are liquid at body temperature but solid at room temperature, whereas oils are liquid at both temperatures.
A triglyceride is made from combining three fatty acids and one glycerol through the process of condensation, meaning that three water molecules are produced. The linkage between each fatty acid and the glycerol is an ester bond. Triglycerides are used as energy stores. The energy from them can be released by aerobic cell respiration.
Fatty
Acids
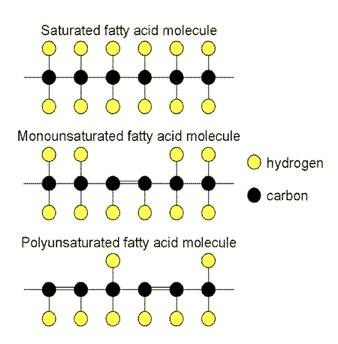
Fatty acids can be saturated, monounsaturated or polyunsaturated
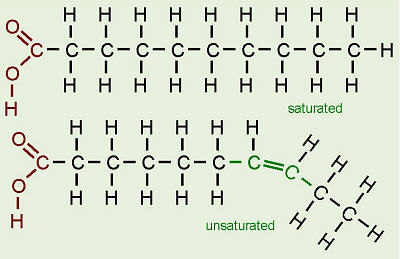
Fatty acids are in the form of a chain of carbon atoms, with hydrogen atoms linked to them by single covalent bonds. It is therefore a hydrocarbon chain. A variable feature of fatty acids is the bonding between the carbon atoms. In some fatty acids all of the carbon atoms are linked by a single covalent bond, while in other fatty acids there are one or more positions in the chain where carbon atoms are linked by double covalent bonds. The difference is that when the carbon atoms are linked by single bonds, they can link with two hydrogen atoms, while if a carbon atom is linked by a double bond, it can only link with one hydrogen atom.
- Saturated fatty acids: these are formed when all of the carbon atoms are linked by single bonds.
- Monounsaturated fatty acids: these are formed when there is only one double bond and the rest are single bonds.
- Polyunsaturated fatty acids: they are formed when there is more than one double bond linking the carbon atoms
Fatty acids can be saturated, monounsaturated or polyunsaturated
Fatty acids are in the form of a chain of carbon atoms, with hydrogen atoms linked to them by single covalent bonds. It is therefore a hydrocarbon chain. A variable feature of fatty acids is the bonding between the carbon atoms. In some fatty acids all of the carbon atoms are linked by a single covalent bond, while in other fatty acids there are one or more positions in the chain where carbon atoms are linked by double covalent bonds. The difference is that when the carbon atoms are linked by single bonds, they can link with two hydrogen atoms, while if a carbon atom is linked by a double bond, it can only link with one hydrogen atom.
- Saturated fatty acids: these are formed when all of the carbon atoms are linked by single bonds.
- Monounsaturated fatty acids: these are formed when there is only one double bond and the rest are single bonds.
- Polyunsaturated fatty acids: they are formed when there is more than one double bond linking the carbon atoms
Unsaturated
Fatty Acids
These can be cis or trans isomers
In these, the hydrogen atoms are nearly always on the same side of the two carbon atoms that are double bonded, which are called cis-fatty acids. The alternative is for the hydrogens to be on opposite sides, which are trans-fatty acids.
The difference between cis and trans fatty acids is that the cis ones have to bend in the hydrocarbon chain at the double bond, which makes the triglyceride’s melting point lower. The trans fatty acids are the complete opposite.
These can be cis or trans isomers
In these, the hydrogen atoms are nearly always on the same side of the two carbon atoms that are double bonded, which are called cis-fatty acids. The alternative is for the hydrogens to be on opposite sides, which are trans-fatty acids.
The difference between cis and trans fatty acids is that the cis ones have to bend in the hydrocarbon chain at the double bond, which makes the triglyceride’s melting point lower. The trans fatty acids are the complete opposite.
Theory of knowledge: There are conflicting views as to the harms and benefits of fats in diets. How do we decide between competing views?.
Cis and Trans fats have been categorized as good and bad. Usually, nutritionists tell their clients to include cis fats, like olive oil, because they can have many positive effects on the body, including weight loss. However, they need to be take with moderation because they are very high caloric components, and if abused, they ingestion can end up in weight gain respectively. Another thing nutritionist might recommend is to stay away from trans or unsaturated fats, not only because they lead to weight gain, but also because they increase the risk of having severe diseases like heart diseases.
Cis and Trans fats have been categorized as good and bad. Usually, nutritionists tell their clients to include cis fats, like olive oil, because they can have many positive effects on the body, including weight loss. However, they need to be take with moderation because they are very high caloric components, and if abused, they ingestion can end up in weight gain respectively. Another thing nutritionist might recommend is to stay away from trans or unsaturated fats, not only because they lead to weight gain, but also because they increase the risk of having severe diseases like heart diseases.