Understanding:
- water molecules are polar and hydrogen bonds from between them.
– Hydrogen bonding and dipolarity explain adhesive, cohesive, thermal and solvent properties of water.
– Substances can be hydrophilic or hydrophobic
Applications:
- Comparison of the thermal properties of water with those of methane. 0 use of water as a coolant in sweat. – Methods of transport of glucose, amino acids, cholesterol, fats, oxygen and sodium chloride in blood in relation to their solubility in water.
Hydrogen bonding in water
Since water molecules are people, meaning that they have a charge, hydrogen bonds form between them
A water molecule is formed by a covalent bond between one oxygen atom and two hydrogen atoms. The bonds between hydrogen and oxygen are unequal in the sharing of electrons because it is a polar covalent bond, meaning that the nucleus of the oxygen atom is more attractive to electrons than the nuclei of the hydrogen atoms
Positively charged particles and negatively charged particles attract each other and form an ionic bond. Water molecules only have partial charges, so the attraction is less but still enough to have significant effects. The attraction between water molecules is a hydrogen bond, which is the force that forms when a hydrogen atom in one polar molecule is attracted to a slightly negative atom of another polar covalent bond.
- water molecules are polar and hydrogen bonds from between them.
– Hydrogen bonding and dipolarity explain adhesive, cohesive, thermal and solvent properties of water.
– Substances can be hydrophilic or hydrophobic
Applications:
- Comparison of the thermal properties of water with those of methane. 0 use of water as a coolant in sweat. – Methods of transport of glucose, amino acids, cholesterol, fats, oxygen and sodium chloride in blood in relation to their solubility in water.
Hydrogen bonding in water
Since water molecules are people, meaning that they have a charge, hydrogen bonds form between them
A water molecule is formed by a covalent bond between one oxygen atom and two hydrogen atoms. The bonds between hydrogen and oxygen are unequal in the sharing of electrons because it is a polar covalent bond, meaning that the nucleus of the oxygen atom is more attractive to electrons than the nuclei of the hydrogen atoms
Positively charged particles and negatively charged particles attract each other and form an ionic bond. Water molecules only have partial charges, so the attraction is less but still enough to have significant effects. The attraction between water molecules is a hydrogen bond, which is the force that forms when a hydrogen atom in one polar molecule is attracted to a slightly negative atom of another polar covalent bond.
Properties
of water
Hydrogen bonds and dipolarity explain the adhesive, cohesive, thermal and solvent properties of water.
- Cohesive property: binding of two molecules of the same type (ex. Two water molecules)
Water molecules are cohesive, this is due to hydrogen bonding. This property allows water transport in plants, since this transportation can only work if the water molecules stick together. Due to the weakness hydrogen bonds, the separation of two water molecules is not rare when the water is being transported within tall trees.
- Adhesive properties:
Hydrogen bonds can happen as well with other polar molecules rather than water. This is the property of adhesion, which is useful in leaves because the water adheres to cellulose molecules in the cell walls.
- Thermal properties:
Water has several thermal properties that are useful to living organisms:
+ High specific heat capacity: the increases of temperature in water require lots of energy because hydrogen bonds need to be broken. To cool down, water must lose relatively large amounts of energy. Water’s temperature remains relatively stable in comparison to air or land, so it is a thermally stable habitat for aquatic organisms
+ High latent heat of vaporization: when a molecule is evaporated, it separates from other molecules in the state of liquid and becomes a vapor molecule. The heat needed to do this is called the latent heat of vaporization. Therefore, evaporation has a cooling effect. Considerable amounts of heat are needed to evaporate water, this is because hydrogen bonds need to be broken. This makes water a good evaporative coolant. Sweating is an example of the use of water as a coolant.
+ High boiling point: Because water has high latent heat of vaporization, it has also has a high boiling point, which is the highest temperature a substance can reach in a liquid state. Water is therefore liquid over a broad range of temperatures from 0 C to 100 C.
- Solvent properties
Only charged substances can be solved by water. This is because the polar nature of water molecules means that they form shells around charged and polar molecules, preventing them from cumpling together and keeping them in solution.
Hydrogen bonds and dipolarity explain the adhesive, cohesive, thermal and solvent properties of water.
- Cohesive property: binding of two molecules of the same type (ex. Two water molecules)
Water molecules are cohesive, this is due to hydrogen bonding. This property allows water transport in plants, since this transportation can only work if the water molecules stick together. Due to the weakness hydrogen bonds, the separation of two water molecules is not rare when the water is being transported within tall trees.
- Adhesive properties:
Hydrogen bonds can happen as well with other polar molecules rather than water. This is the property of adhesion, which is useful in leaves because the water adheres to cellulose molecules in the cell walls.
- Thermal properties:
Water has several thermal properties that are useful to living organisms:
+ High specific heat capacity: the increases of temperature in water require lots of energy because hydrogen bonds need to be broken. To cool down, water must lose relatively large amounts of energy. Water’s temperature remains relatively stable in comparison to air or land, so it is a thermally stable habitat for aquatic organisms
+ High latent heat of vaporization: when a molecule is evaporated, it separates from other molecules in the state of liquid and becomes a vapor molecule. The heat needed to do this is called the latent heat of vaporization. Therefore, evaporation has a cooling effect. Considerable amounts of heat are needed to evaporate water, this is because hydrogen bonds need to be broken. This makes water a good evaporative coolant. Sweating is an example of the use of water as a coolant.
+ High boiling point: Because water has high latent heat of vaporization, it has also has a high boiling point, which is the highest temperature a substance can reach in a liquid state. Water is therefore liquid over a broad range of temperatures from 0 C to 100 C.
- Solvent properties
Only charged substances can be solved by water. This is because the polar nature of water molecules means that they form shells around charged and polar molecules, preventing them from cumpling together and keeping them in solution.
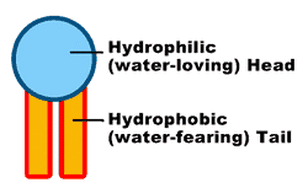
Hydrophilic
and hydrophobic
Substances can be hydrophilic or hydrophobic
- Hydrophilic substances: these substances are attracted to water. All substances that dissolve in water are hydrophilic, as well as substances that water adheres to.
- Hydrophobic substances: these substances repel water; they are insoluble in water specifically. Molecules are hydrophobic if they do not have negative or positive charges and are nonpolar.
Substances can be hydrophilic or hydrophobic
- Hydrophilic substances: these substances are attracted to water. All substances that dissolve in water are hydrophilic, as well as substances that water adheres to.
- Hydrophobic substances: these substances repel water; they are insoluble in water specifically. Molecules are hydrophobic if they do not have negative or positive charges and are nonpolar.
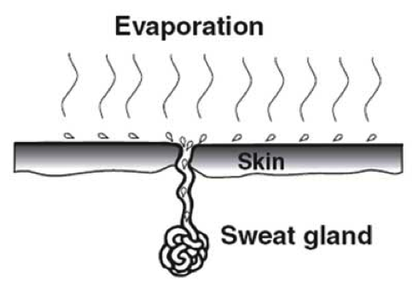
Cooling
the body with sweat.
The glands in the skin secrete sweat. The sweat is carried out by narrow ducts on the surface of the skin where it spreads out. The heat needed for the evaporation of water in sweat is taken from the tissues of the skin, reducing their temperature. Blood flowing through the skin is therefore cooled. This is an effective method of cooling the body because water has a high latent heat of vaporization.
The glands in the skin secrete sweat. The sweat is carried out by narrow ducts on the surface of the skin where it spreads out. The heat needed for the evaporation of water in sweat is taken from the tissues of the skin, reducing their temperature. Blood flowing through the skin is therefore cooled. This is an effective method of cooling the body because water has a high latent heat of vaporization.
Theory of knowledge: Claims about the “memory of water” have been categorized as pseudoscientific.
What are the criteria that can be used to distinguish scientific claims from pseudoscientific claims?
The word pseudoscientific stands for something that cannot yet be categorized as completely scientific. In order for something to be catalogued as scientific, coherent and reasonable research has to be done and the possible hypothesis need to be proven with reliable and proved evidence done by running tests.
What are the criteria that can be used to distinguish scientific claims from pseudoscientific claims?
The word pseudoscientific stands for something that cannot yet be categorized as completely scientific. In order for something to be catalogued as scientific, coherent and reasonable research has to be done and the possible hypothesis need to be proven with reliable and proved evidence done by running tests.